A link to life
-
- from Shaastra :: vol 05 issue 03 :: Mar 2026
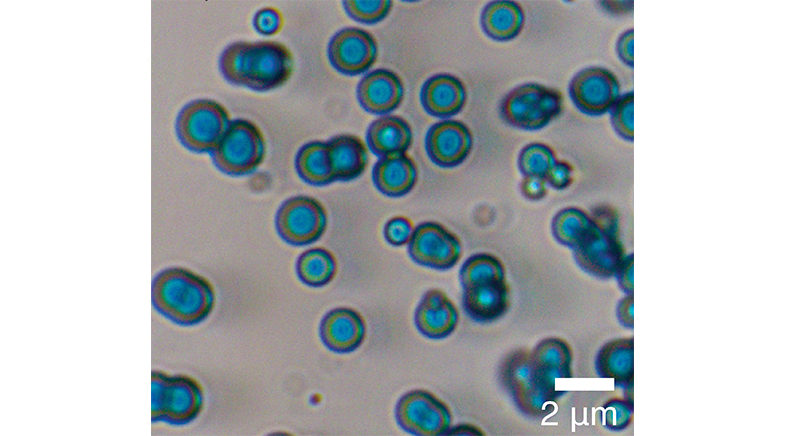
'Protocells' synthesised from a chemical cocktail could bridge a gap in origin-of-life studies.
Shashi Thutupalli knew himself to be a practising physicist working mainly on active matter — an area that includes the study of murmurations in birds, self-propelling parts of the cell, and coordinated movements of fish. A chance encounter led him to a field that nestles in the interface of physics, biology and chemistry — prompting him to try and synthesise lifelike particles out of simple chemicals.
It began when the National Centre for Biological Sciences (NCBS) physicist read The Principles of Life by Hungarian chemist Tibor Gánti back in 2019. From the book, Thutupalli learnt about the work of two chemists at the University of Allahabad — S. Ranganayaki and Krishna Bahadur, who worked on synthesising protocells out of simple chemicals in the 1950s. Bahadur had given the name Jeewanu (particles of life) to the entities. The compelling chemical logic behind the work piqued Thutupalli's curiosity. His PhD student, Nayan Chakraborty, too, was inspired by the story, and applied himself to this effort.
Thutupalli and Chakraborty have come up with research that could bridge a longstanding gap in the origin-of-life studies. By merely tuning chemical parameters such as pH and using molybdenum salts as a catalyst, they have created in the lab what they call "protocells", precursors to living cells, from four simple molecules. These protocells are compartments that grow, metabolise, and have a propensity to replicate themselves in a self-sustaining manner.
They show rudiments of metabolism, grow over time, and contain smaller microspheres, which, when released, grow in size.
The question of how life began on Earth boils down to this: how did simple non-living molecules and their chemistry transform into self-sustaining, evolving, and reproducing cells? The answer is in two parts with a missing connection. In the 1950s, chemists Stanley Miller and Harold Urey showed that individual building blocks of life, such as amino acids, sugars, and nucleotides, can be made from simple starting materials. On the other hand, biologists and biophysicists have shown that if you start with sufficiently complex biomolecules, you can assemble them into cell-like structures. But there is a "continuity" gap between these two efforts. How can chemistry travel from a simple feedstock to forming a functional, self-organising compartment — or a protocell — in one continuous process? Thutupalli's group sought to address that gap.
NEW LEASE OF LIFE
After three years of study, Thutupalli and Chakraborty have arrived at a minimalist solution. Their finding is posted as a preprint and is under peer review. They mixed four chemical compounds composed of small molecules (formaldehyde, ferrous sulphate, diammonium molybdate, and diammonium hydrogen phosphate). These contain the elements necessary for life: carbon, hydrogen, nitrogen, oxygen, phosphorus and sulphur, with iron and molybdenum acting as catalysts. These were in a solution with a variable pH or acidity index. When they hit the right proportions and pH value, blue compartments, or microspheres, formed, which Thutupalli calls protocells. Remove any one of the set of chemicals, and these microspheres do not form.
Further, these compartments show a rudimentary form of metabolism, involving chemical processes that generate energy and heat; they grow in size over time; and contain smaller microspheres that, when released into the solution by rupturing the shell, themselves grow in size, indicating that the microspheres exhibit the potential for multiplication. These are three characteristics of living beings: metabolism, growth and replication.

"One reason why this work is impactful is that it reached out to a line of work that was somehow lost, even though it was quite productive," says NCBS physicist Mukund Thattai, who was not associated with Thutupalli's research. He points out that in the 1950s, Bahadur and Ranganayaki were sharply criticised, and their results were downplayed as mere artefacts. The NCBS work emphasises that the outcomes reached by the Allahabad University chemists were not artefacts. "They (Thutupalli and Chakraborty) have gone further and made the recipe with even fewer molecules, and arrived at a minimal set," Thattai says. He refers to the simplicity of the requirements — ambient conditions, except for a low pH (of 2), which corresponds to a high concentration of hydrogen.
A protocell is a precursor to a cell, a structure that exhibits some but not necessarily all the properties of a living cell.
The work was arduous: Chakraborty had to prepare hundreds of individual reaction mixtures, varying the concentrations of each of the four starting compounds and the pH of the solution. This meant setting up arrays of reactions in deep-well and multi-well plates, each well containing a different combination of reagent concentrations. "Nayan really led the way… (with) painstaking, methodical experimental work," says Thutupalli.
It took three years to get what was a chemical phase diagram — showing which combinations of conditions produce microspheres, which produce amorphous aggregates, and which remain clear solutions.
METABOLISM AND GROWTH
Using high-resolution imaging, including scanning and transmission electron microscopy, Chakraborty and Thutupalli showed that these microspheres had well-defined shells. The compartments would stick to each other when they collided, but not merge as two droplets of mercury would. The researchers showed that the interior of these compartments was distinct from their exterior.
To see whether the mixture showed rudimentary metabolic activity, they measured its temperature and found it emitted heat over and above that required by the chemical reactions alone for up to 21 days. This was not the case with a control mixture from which formaldehyde had been removed.
When they ruptured the shell of the round bodies 12 hours after they were formed, smaller spherical structures emerged. These grew into larger spheres and were similar to the spheres they came from. They conclude this could be a rudimentary form of propagation.
The team also found that the microsphere-enriched solution had the chemical signatures of complex carbon-containing compounds, identified by ratios of constituents (stoichiometric ratios) as lipid-like, amino-acid/peptide-like or carbohydrate-like. These are the building blocks of complex molecules such as RNA, DNA, and proteins. "It has never been shown before that you can make all these in the same pot, under the same conditions," says Thattai. While he stops short of calling it a proof of the existence of macromolecular compounds — for a proof demands going beyond mass spectroscopy — he holds that these are "very strong evidence" of chemical signatures.
BINDER AND CATALYST
The catalyst molybdenum plays a key role. The element has the property of bringing together molecules to form spherical structures. "The catalysis that's happening here is rather sophisticated," Thattai says, referring to the possible propagation of small spherical structures. "What they don't have is a cell," Thattai adds. A cell has a tight membrane which separates the inside from the outside. "Without a membrane, Darwinian evolution is not possible. It's not clear that has happened here," he says.
However, Thutupalli does not call it a cell but a protocell. According to him, to qualify as a protocell, the spherical compartment does not require a lipid membrane. A protocell is a precursor to a cell, a structure that exhibits some but not necessarily all the properties of a living cell. The key features are compartmentalisation, sustained chemical activity, and some form of growth. "Our structures exhibit all three," he says.
Molybdenum-rich microspheres that closely resemble their laboratory protocells have recently been discovered inside marine sponges in the Indo-Pacific Ocean. To Thutupalli, this convergence suggests that the chemistry they observe in the laboratory may be something that nature does on its own, even today, in the right geochemical environments. "We want to be careful about the interpretation: we are not claiming that the sponge structures are protocells, or that they formed by the same process. But the convergence is provocative," he says.
Sudha Rajamani, a biologist from the Indian Institute of Science Education and Research Pune who works on origin-of-life questions, describes the NCBS research as "very exciting" work. "It is extremely important to have a multipronged approach when it comes to understanding something as fundamental as the transition from chemistry to biology on early Earth," she says. The focus of early experiments has largely been on chemical and geological or geochemical aspects. "(Thutupalli) brings in the very interesting and important flavour of understanding the physics of lifelike systems," she says.
See Also:
Have a
story idea?
Tell us.
Do you have a recent research paper or an idea for a science/technology-themed article that you'd like to tell us about?
GET IN TOUCH














