Cost-effective carbon capture
-
- from Shaastra :: vol 02 issue 02 :: Mar - Apr 2023
The global temperature is now 1.1°C higher than in the pre-industrial era. It's a no-brainer that if that is not to cross the 1.5° limit, the amount of carbon dioxide (CO2) emissions needs to be drastically reduced.
One way to achieve this is to cut dependency on fossil fuels and increase the use of renewable energy. An equally, if not more, effective way is to convert the CO2 emitted as an effluent into products. Research groups around the world have developed innovative ways to do so, but these are far from being cost-effective.
The photocatalytic technique has yield rates that are 180 times higher than those of similar systems developed to date.
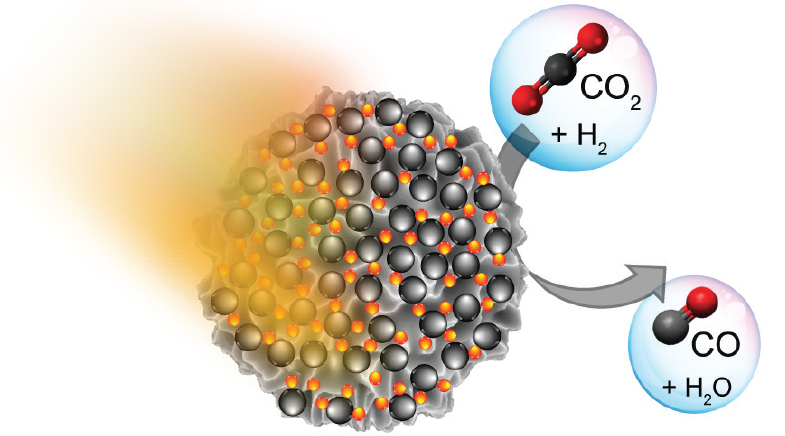
A team of chemists led by Vivek Polshettiwar, Professor of Chemical Sciences at the Tata Institute of Fundamental Research (TIFR) in Mumbai, may have cracked this conundrum. In a study (bit.ly/photocatalyst-paper) published in the journal ACS Nano in February this year, the team synthesised a new photocatalyst that can efficiently convert CO2 into carbon monoxide (CO), which when mixed with hydrogen can produce chemicals and fuels that are used commercially today. The photocatalytic technique, which uses temperatures in the 80-220°C range and normal pressure, has yield rates that are 180 times higher than those of similar systems developed to date.
The catalyst produced over 55 litres of CO per hour and demonstrated the tenacity to work for 100 hours at a stretch. Polshettiwar is confident that it can stay active for years, working 24x7, with some regeneration steps in between.
"The work is significant because of the yield rate as well as the quality of the catalyst that they developed," says Ramanathan Vaidhyanathan, Professor of Chemistry at the Indian Institute of Science Education and Research Pune, who was not involved in the study.
The TIFR scientists synthesised the novel photocatalyst, named black gold-Ni, by loading small gold nanoparticles onto fibrous nanosilica. The scientists named the material 'black gold' because of its shining nature and spiked it further with nickel nanoparticles to produce the photocatalyst.
The scientists followed this up further and developed a new technology to capture CO2 from effluents using carbon nanospheres. The work done by Polshettiwar's team, in association with researchers at the Bhabha Atomic Research Centre, was published in Langmuir journal in March (bit.ly/langmuir-paper).
Have a
story idea?
Tell us.
Do you have a recent research paper or an idea for a science/technology-themed article that you'd like to tell us about?
GET IN TOUCH














