Eyes on the light
-
- from Shaastra :: vol 05 issue 02 :: Feb 2026
Optogenetics, once confined to discovery science, enters into therapeutics, offering solutions to a range of conditions.
The year 2019 was one of hope for four visually impaired patients at the JPM Rotary Club of Cuttack Eye Hospital and Research Institute in Odisha. They had been chosen for a new therapy that had the potential to restore their vision. The therapy had been tested only in animals till then, but the patients were desperate and willing to give it a shot.
Santosh Mahapatra, an ophthalmologist at the hospital, was familiar with the everyday frustrations and challenges faced by his visually impaired patients. His search for possible treatments for his patients led him to the work of Samarendra Mohanty, a U.S.-based researcher who was developing optogenetics-based therapy for genetic diseases of the retina. The therapeutic approach used gene therapy to insert light-sensitive proteins – called opsins – into surviving retinal cells in order to make them sensitive to light and act as photoreceptors, which are retinal cells that perceive light. Opsins act as artificial photoreceptors in retinal cells that have lost the photoreceptor function due to a genetic abnormality.
Mahapatra hoped that the optogenetics-mediated resurrection of photoreceptors would help his patients. He approached Mohanty to conduct an investigator-initiated pilot trial at his hospital. Each of the four patients willing to enrol for the trial received the therapy as a single injection in their eyes. Each dose consisted of copies of an AAV (adeno-associated virus) — a vehicle to deliver the synthetic opsin gene into the retina. The delivery vehicle enters the cell and forms an episome (remains separate from the main host DNA in the nucleus). It does not integrate into the genome, but remains stable inside the nucleus and expresses protein when activated.
Optogenetics introduces genes encoding light-sensitive proteins called opsins into targeted cells, making them responsive to light.
Mohanty and his colleagues in the U.S. had carefully developed this synthetic opsin – also called synthopsin – to fit right into the role of a human photoreceptor. 'It is actually a fused protein taken from three different microbial organisms,' says Mohanty, Co-Founder and Chief Scientific Officer of the U.S.-based Nanoscope Therapeutics, which developed the therapy.
Unlike other commonly used lab-developed opsins that are sensitive to specific wavelengths of light, the one developed by Mohanty's team is sensitive to a broad range of wavelengths from blue to red. It does not require high-intensity LEDs for activation, only ambient light, and has a quick reaction time. 'We had to make it very sensitive, fast, and then also broadband. So those three unique properties were engineered into the opsin,' says Mohanty.
The four patients at the Cuttack hospital were observed for one year; all of them showed significant improvement in vision and mobility. The results of the trial were published (bit.ly/optogeneticstrial) in May 2025 in the journal Molecular Therapy. Following the pilot trials in Odisha, Nanoscope conducted a randomised, controlled Phase 2b trial in the U.S., in which patients showed a statistically significant improvement in vision over the two-year study period. 'Some were able to ride a bicycle, some were able to go back to work, and play with their grandkids,' says Mohanty.
SHINING LIGHT ON NEURAL CIRCUITS
Optogenetics has been the tool of choice since the early 2000s for researchers studying the brain. An elegant method to identify neural circuits associated with specific behaviour and diseases, optogenetics adopts a simple strategy – introducing genes encoding light-sensitive proteins called opsins into targeted cells, making them responsive to light. When light of a specific wavelength is delivered, the cells are activated or inhibited – almost instantly – allowing researchers to study how neural circuits control behaviour and disease with high precision.
Introducing light-sensitive proteins into specific cells in humans and delivering light into deep tissue are challenging.
Arnab Barik, Assistant Professor at the Centre for Neuroscience, Indian Institute of Science, Bengaluru, uses optogenetics (and other tools) to study the neural circuits involved in pain and itch. He is particularly interested in the brainstem circuits at the base of the brain, which receive pain-related inputs from the spinal cord and relay them to higher brain regions for further processing. The pain circuits also communicate with stress and anxiety circuits; this communication is bidirectional – chronic pain can worsen anxiety and stress, and chronic stress can worsen pain. Barik and his colleagues are working to identify the specific circuits involved in the interaction through their studies in mice. A region called the lateral hypothalamus, they have found, acts as a key node in transmitting stress-related information to the brainstem.
Hasan Mohammad, Assistant Professor at the Indian Institute of Science Education and Research (IISER) Mohali, relies on optogenetics to study neural circuits involved in addiction and compulsive eating behaviour. Mohammad is particularly interested in the habenula, the brain region associated with aversion behaviour; it connects to the hypothalamus, where feeding-related neurons are located. Targeting these neural circuits may offer new strategies to treat compulsive eating disorders, he observes.
LIGHT-BASED THERAPEUTICS
Studies like those being conducted in the labs of Barik and Mohammad have increased in numbers over the past decade. It has become easy to use optogenetics in mice to study the neural circuits underlying fear, pain, motivation, and a host of other behaviours and diseases.
The foray of optogenetics into therapeutics, though, has taken relatively longer. Introducing light-sensitive proteins into specific cells in humans and delivering light into deep tissue are challenging. But the eye is unique: it is naturally exposed to light, so no deep-brain implants or light sources are needed, thereby removing one of the biggest barriers to optogenetic therapy. Moreover, the eye is a well-enclosed organ, which makes precise gene delivery easier and safer compared to the rest of the brain. Which is why therapies such as those developed by Nanoscope are becoming possible (see 'The eyes have it').
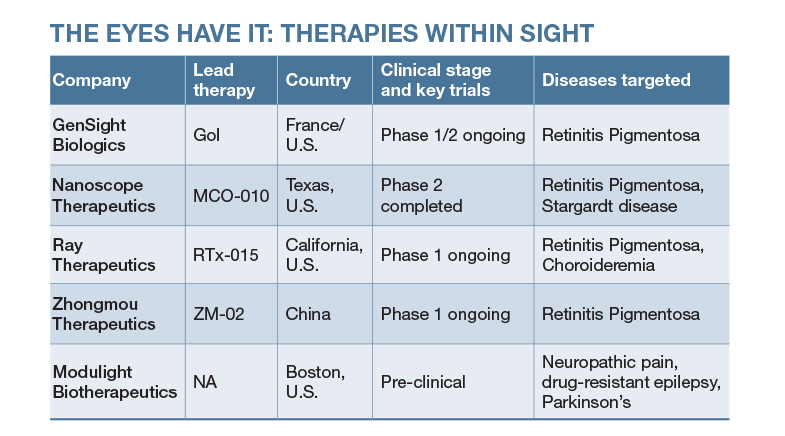
The range of conditions that optogenetics can help treat is slowly expanding as well. Researchers are figuring out ways to treat conditions such as chronic pain and epilepsy with optogenetics. It could be a good tool for skin-related problems, too, reckons Mohammad. 'I don't know if people are going in that direction, but it's certainly doable,' he says. Optogenetics, he adds, can be used to treat conditions in body parts with good access to light, such as ears or teeth.
Optogenetics could be an efficient treatment tool for neurological diseases where electrical stimulation is a therapeutic strategy. 'There are a lot of opportunities with optical stimulation that can completely go above and beyond what exists with electrical stimulation now. Even for rehabilitation, if we can use opsins in the spinal cord, we might be able to do rehabilitation without any electrical stimulation, which can cause pain as a side effect,' says Sapna Sinha, Postdoctoral Fellow at the Massachusetts Institute of Technology, U.S., who works on developing opsins better suited for human therapeutic usage. Her work focuses on designing and delivering the opsin gene into the human body so that it is tolerated well by the immune system and retains its functionality, and can enable the use of optogenetic therapy for a wider variety of applications.

Mohanty is part of another start-up, Opsin Biotherapeutics, which is developing optogenetic therapies for chronic pain. The team aims to make the pain-blocking neurons in the spine sensitive to light so they can be activated by simple LED light.
Widespread use of optogenetic therapies in clinics, however, remains some time away. But the success of early trials shines a light on the potential of precisely engineered light in treating ailments.
Have a
story idea?
Tell us.
Do you have a recent research paper or an idea for a science/technology-themed article that you'd like to tell us about?
GET IN TOUCH














